|
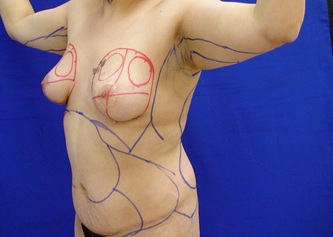
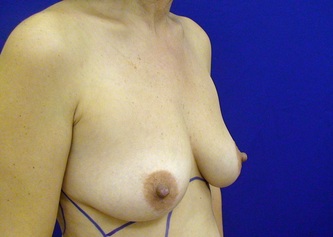
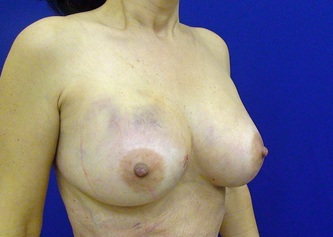
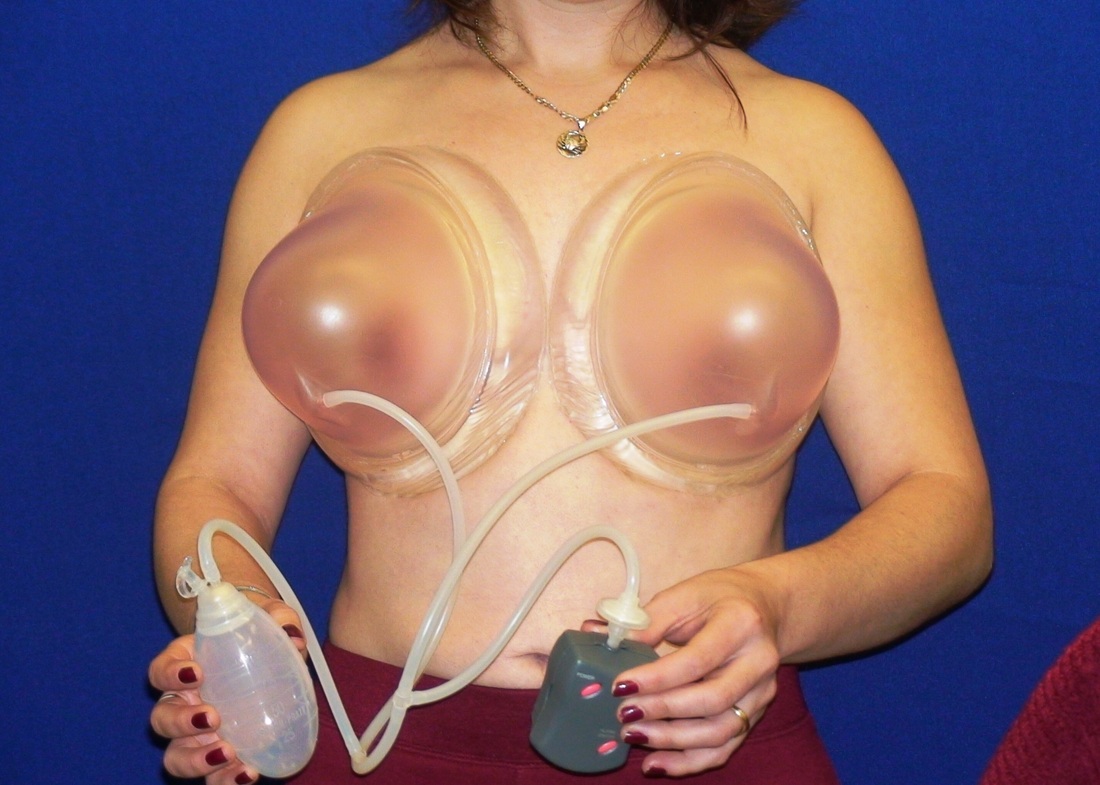
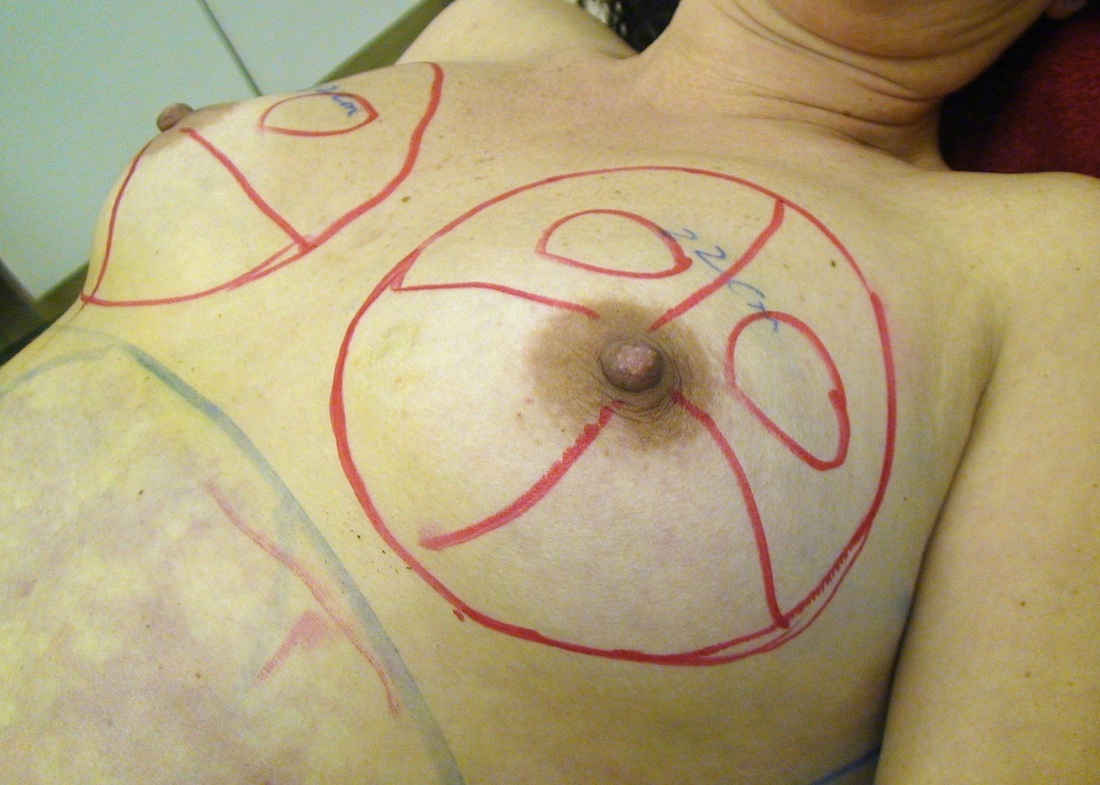
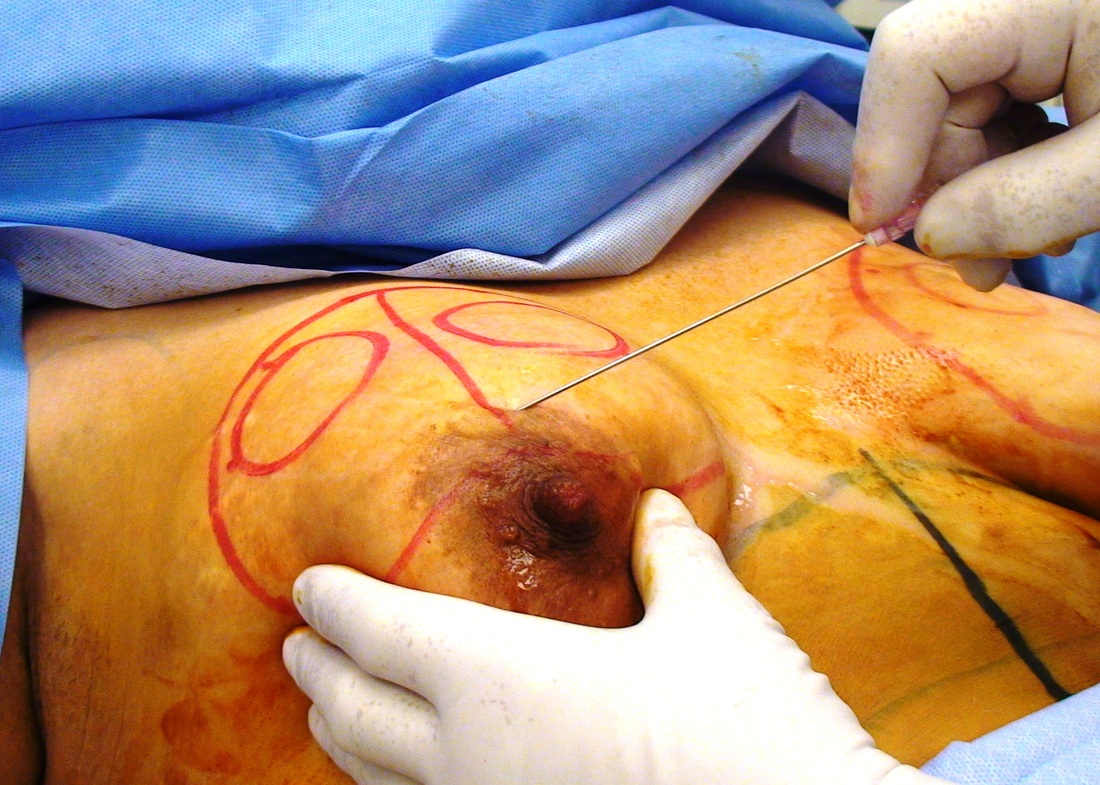
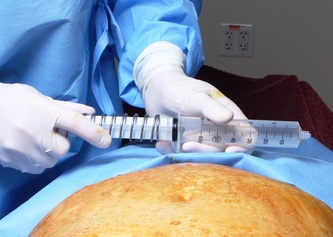
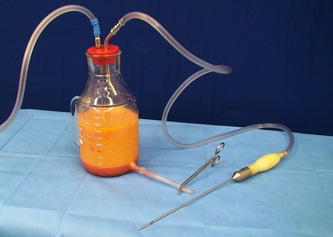
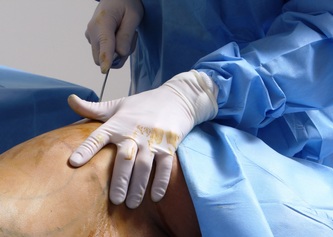
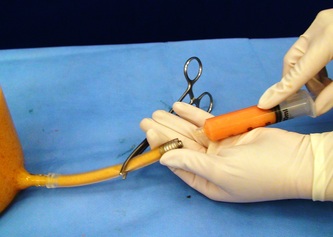
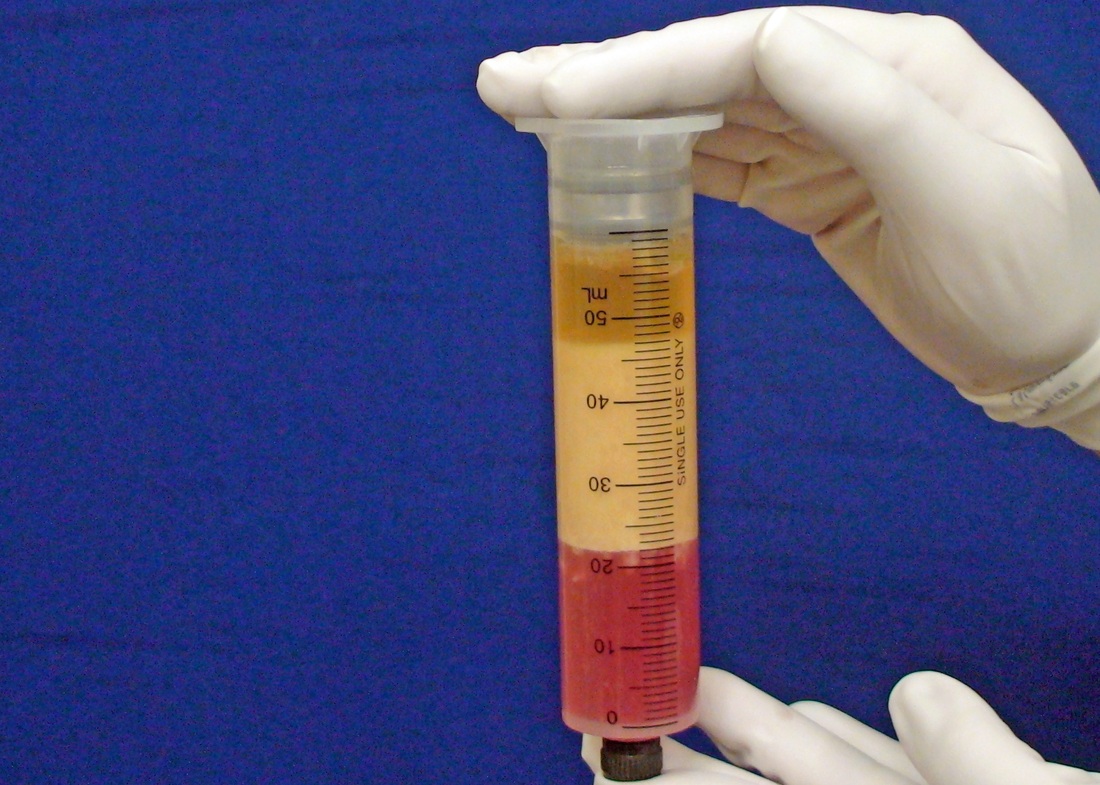
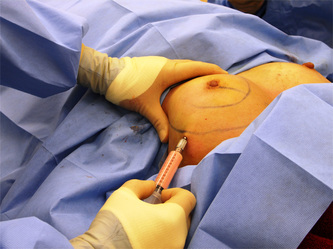
This was the topic of a recent lecture I gave at the American Society of Cosmetic Breast Surgery annual meeting in Newport Beach, California. The slide show sans narration is available here. It is also the topic of an upcoming chapter in a book edited by Melvin Shiffman. IntroductionBreast augmentation with injections of autologous fat harvested by liposuction was first described in the mid 1980s (2). Early enthusiasm worldwide was soon tempered by concern, albeit speculative, that such transplants would confound or delay the radiologic diagnosis of breast cancer (3). Ongoing critical analysis over recent years has demonstrated, however, that these concerns do not appear to be heightened in comparison to other cosmetic breast procedures and these findings have stimulated a renewed interest in this type of breast augmentation (4,5). To date, this procedure appears best-suited for small-volume augmentation and for cosmetic tissue remodeling around existing synthetic breast implants and sometimes requires multiple treatment sessions to achieve a desirable lasting result (Figure 1). The terms lipofilling, lipomodeling, and lipoinjection are synonyms used to describe autologous fat transfer. Structural fat grafting refers to a specific technique of autologous fat transfer (4). IndicationsThere are two indications for breast augmentation: (1) a cosmetic desire to have larger breasts, and (2) reconstruction of congenital or acquired breast deformity or absence. The best candidates for breast augmentation by any modality are nonobese women displaying relatively symmetrical breasts, a symmetrical frame and lacking breast ptosis. Ptosis is evaluated with the patient standing and is defined as descent of the center of the nipple-areola complex (NAC) below the level of the inframammary fold (IMF). Grading systems are described elsewhere (6). The best candidates for cosmetic breast augmentation by autologous fat transfer are those seeking a modest increase in size, who (1) have adequate fat deposits for harvest by liposuction, (2) understand that an unpredictable proportion of the transplanted fat and associated volume, will resorb during the healing phase, (3) are aware that multiple sessions may be required to achieve a suitable size increase, and (4) are aware that saline or silicone implants are options that deliver predictable size augmentation and they reject those options. A subgroup of patients requesting breast augmentation with autologous fat are primarily interested in liposuction body contouring. Their motivations for breast augmentation lie more in the financial convenience of combining the body contouring procedure with autologous fat transfer and are less interested in attaining a specific increase in breast size. Nonetheless, they should always be counseled regarding the limitations of the breast augmentation with autologous fat and of the other common breast augmentation options. The correction of contour irregularities around existing saline or silicone breast implants is a less common, but effective application of autologous fat transfer to the breast. It is prudent in such cases to be fully prepared to perform an implant exchange at surgery in the event of accidental implant perforation. Rippling and upper pole volume deficiencies respond well to fat transplantation. Capsular contracture has been reported to improve after fat grafting around the constricted capsule (4). Screening & EvaluationGeneral screening criteria for liposuction, then specific criteria for breast surgery are employed. Since breast augmentation by autologous fat transfer typically generates only a modest increase in breast volume, it essential to determine that the patient is not seeking a larger increase in breast volume. Otherwise, the patient will be dissatisfied with the result and with the surgeon for not offering an alternative better suited to her desires. The ideal cosmetic breast patient has no breast symptomatology, pathology, or risk factors for breast disease. Breast specific screening begins with a thorough history and family history followed by a complete physical examination of the breasts with attention to any masses or lymphadenopathy. A history of breast cancer treated by conservative resection is an absolute contraindication to the procedure. Mammography is performed on patients meeting either medical diagnostic or routine screening criteria. Some surgeons prefer to screen all cosmetic breast patients with mammography prior to surgery because of concern that postsurgical changes may obscure subsequent breast cancer screening. All patients are counseled that cosmetic breast surgery may produce calcifications, cysts or palpable lumps that may require diagnostic imaging and interventions including biopsy and can never be assumed to be benign. Medications, supplements, herbs and other substances with the capacity to impair coagulation should be discontinued in advance of surgery. Substances which interact negatively with anesthetic agents and perioperative medications should also be withheld. If they cannot be discontinued or substituted, the surgical plan will need to be modified, delayed or withheld. Cigarette smoking is not a contraindication to liposuction nor to autologous fat transfer, but smokers typically display an atrophic dermis and less skin elasticity which both increase the likelihood of postoperative skin wrinkling. They also demonstrate greater resorption of transplanted fat. Photography of the breasts prior to surgery, before and after marking their measurements, completes the preoperative process. Written release forms specific to photography are mandatory. A routine that includes frontal, oblique and lateral views with arms both at rest and elevated provides an accurate record of asymmetries and of any preexisting scars or contour deformities. External Tissue ExpandersBreast augmentation with external tissue expanders was introduced in 1999 (7). The method, currently known as the Brava system, utilizes a pair of self-sealing rigid polyurethane domes which are applied directly over the breasts to create a negative pressure which causes tissue distraction which, in turn, stimulates tissue growth. Although the technology is effective, it has not gained popularity because it requires a minimum of ten hours of continuous daily use for a minimum of ten weeks to produce results and the results, under the best of circumstances, are typically less than a one-cup size augmentation. More recently, however, the Brava device has been utilized as a means of expanding the superficial planes of the breast in preparation for autologous fat transfer allowing for more fat to be injected under less pressure. The protocol under study involved 4 weeks of pre-operative Brava use for 10 hours daily followed by postoperative use as a shape-supporting stent for the first 7 days (8). Adipose-Derived Stem & Stromal Cells (ADSCs)Multipotent stem cells present in adipose secrete potent growth factors which have been shown to enhance tissue survival in certain settings (9). These findings have stimulated novel techniques of autologous fat transfer that enrich the concentration of stem cells in the harvested fat in preparation for injection (10). Theoretical benefits of adding ADSCs to the harvested fat include: (1) ADSC differentiation into adipocytes, (2) ADSC differentiation into endothelial cells with subsequent increase in blood supply, (3) ADSC release of angiogenic growth factors, (4) ADSC-mediated protection from graft ischemia, and (5) accelerated wound healing (9). Theoretical concerns that ADSC-enriched grafted fat might stimulate the development of breast cancer in vivo have not been supported by clinical findings to date (11). Presently, the use of ADSC-enriched fat in breast augmentation remains in its infancy and there is no clinical data to suggest that it outperforms non-enriched fat in vivo. Platelet-Rich Plasma (PRP)Recent commercial marketing of platelet-rich plasma (PRP) as an aesthetic filler and as an agent with the potential to enhance “handling characteristics of autograft” (12) has prompted the development of multiple techniques and applications of PRP-enriched autologous fat. PRP contains growth factors active in the initiation of wound healing which, in theory, might prove beneficial to the survival of grafted fat. However, the only data currently available relates to in vitro studies (13). A recent small retrospective review of 42 women augmented with autologous fat, 25 of whom received PRP-enriched fat, demonstrated no effect of PRP enrichment on aesthetic outcomes or rates of liponecrosis (14). In our experience, we find that the addition of PRP to centrifuged fat in a 1:10 ratio thickens the consistency of the fat and helps in maintaining the contours of the recipient site in the immediate postoperative period whether it be breast or other sites. Surgical MarkingsThe breasts are marked with the patient standing and the arms down. The margins of the breast mound are marked circumferentially. The breast is divided into quadrants with radial lines drawn from the areolar edge to the margins of the breast mound at the 12:00, 3:00, 6:00, and 9:00 positions. Areas within these quadrants requiring extra volume are then marked and photographed. Operative TechniqueBreast augmentation by autologous fat transfer and fat harvesting by liposuction is typically performed under tumescent local anesthesia (TLA) with or without supplementary intravenous sedation; the latter is employed to calm unusually anxious patients and is conducted by an anesthesiologist. General anesthesia is unnecessary because TLA results in total anesthesia of the targeted tissues. However, techniques which also target fat injection into the pectoralis major muscle utilize general anesthesia (4). Broad spectrum prophylactic antibiotics targeting skin flora are administered intravenously immediately prior to surgery and the patient is positioned in the supine position with the arms away from body. Continuous noninvasive monitoring of blood pressure, pulse oximetry and 3- or 5-lead EKG are instituted. The skin is scrubbed with chlorhexidene or betadine and latex free drapes are applied. The procedure begins with TLA infiltration of the harvest areas and of the breasts. The harvest sites are infiltrated to a tumescent level of tightness by standard techniques described elsewhere (15). TLA is administered via infusion pump and a long spinal needle (18G x 6”, Quincke tip). TLA concentrations per liter of normal saline are as follows: lidocaine 750mg, epinephrine 1mg, sodium bicarbonate 10mEq (8.4% x 10ml). The total lidocaine dose should not be allowed to exceed 50mg/kilogram of body weight. We prefer sharp spinal needles over reusable blunt-tipped infiltration cannulas because (1) they pass through the dense breast connective tissue with far less resistance and tissue drag which results in smoother and more precise guidance by the surgeon, and (2) by the nature of their design, spinal needles deliver the anesthetic fluid wave ahead of the sharp instrument tip thereby minimizing any painful needle tip sensation by the patient. TLA infiltration of the breast in preparation for autologous fat transfer is distinct: A low-volume infiltration of TLA is administered superficially over the entire breast mound approximately 1-cm in thickness. A deeper infiltration is not carried out because the autologous fat is injected superficially. A larger volume infiltration will distort the tissues and impede the accuracy of the augmentation. TLA infiltration begins in the center of the breast mound at the 12:00, 3:00, 6:00, and 9:00 positions at the areolar margin. The skin at these sites is anesthetized with a small wheal of TLA delivered through a small needle (30G) prior to inserting the spinal needle. The spinal needle is then advanced parallel to the skin changing direction in a fan pattern at a low infusion rate. Once the breasts and the harvest sites have been anesthetized adequately, liposuction fat harvesting is carried out using standard technique (15). Cannulas of 3-mm diameter or narrower are preferred because they deliver small fat parcels which are easy to inject through narrow fat transfer cannulas and produce a smoother fill. Fat is collected in either a syringe or in a sterile large-volume glass canister connected to the working cannula via sterile tubing and to the liposuction aspirator pump via a second sterile tubing line. It is important to keep the aspirator suction pressure low (300-330mm Hg) during harvesting to minimize barotrauma to the adipocytes. Centrifugation of the aspirated fat helps to remove blood and excess fluid in order to achieve a more accurate fill of the breast. Centrifugation is carried out in 10-cc syringes at 3,000 rpm for three minutes; oils and fluids are decanted from the top and bottom of the syringe, respectively Centrifugation typically results in the reduction of the apparent volume of fat in the lipoaspirate by one half. Thus, we target a fat volume collection endpoint of twice the desired fill volume. Since we usually inject a maximum of 200ml of fat per breast, we seek to collect 400ml of fat per breast in the lipoaspirate whenever feasible. Naturally, in leaner patients this target may not be reached. The centrifuged fat is transferred to small syringes in preparation for injection. We prefer 5-10cc syringes. Blunt-tipped 14G x 15cm injection cannulas with single side ports (Coleman Type II) are attached to the syringes for the injection process. Injection is carried out superficially in a fan-like pattern through the same points on the areolar margin (12:00, 3:00, 6:00, 9:00) through which the spinal needles were inserted during TLA infiltration. In most instances, the needle track is sufficient to admit the cannula; if not, a 1-mm dermapunch incision is made. When necessary, additional injections are made along the periphery of the breast margin. The injection cannula is inserted to reach the level of the breast margin marks and fat is injected evenly as the cannula is withdrawn. Typically 1ml of fat is deposited per pass. Large volume deposits are avoided in order to maximize contact with of the transplanted fat with the native recipient tissues. In the event that contour deformities or tissue distortion resist volumetric expansion, tension in the injection planes is released by focal undermining with V-tipped dissection cannulas, disruptor cannulas or microscissors The injection process can be done with the patient supine or with the table flexed and the patient in the semi-recumbent position or they can be alternated as needed to assess the contours as they develop. It should be noted that current techniques of autologous fat transfer to the breast intentionally avoid direct injection into the breast parenchyma. While the injection of the superficial planes usually gives sufficient size enhancement, some surgeons seeking to achieve increased breast mound projection target additional injections into the pectoralis major and the prepectoral layers. Post-Op ManagementAt the conclusion of the procedure, the areolar injection points are covered with steri-strips. Absorbent pads and compression garments are placed over the harvest sites. The breasts are fitted with a tight conforming bra and the patient is discharged home. The patient is seen for follow-up at 24-48 hours post-surgery primarily for surveillance of the harvest sites. Active fluid expression of these areas, if necessary is conducted by the nursing team. The breasts are examined for signs of excess bruising or infection; the latter is rare. Oral broad spectrum antibiotic coverage for skin pathogens is maintained for the first seven days after surgery. Analgesic needs are typically minimal and NSAIDs are usually sufficient. Patients are able comfortably to return to a desk job by 48 hours post-surgery. Additional postoperative surveillance is conducted on a bi-weekly, then monthly basis as deemed appropriate. The final breast volume stabilizes by approximately three months after surgery. ComplicationsDamage to the chest wall is unlikely with autologous fat transfer to the breast because the injection is limited to the superficial plane. Of more concern, but rare, would be infection of the transplanted fat. We have not seen this complication on the breast, but in other areas such as the buttocks, infection of transplanted fat is managed by the expression of the transplanted fat through the sites of injection either manually or with the assistance of liposuction cannulas in conjunction with aggressive antibiotic coverage and may require more than one session. Significant loss of augmented breast volume is common especially in cigarette smokers. The patient needs to be counseled thoroughly prior to surgery that a noticeable portion of the immediate postoperative fill volume represents anesthetic fluid within the breast and fluid within the transplanted fat. Interventions which promote the healing process include adequate caloric intake and smoking cessation, but these no offer no guarantee of superior results. Repeat sessions of autologous fat transfer to the breast should be scheduled at an interval that allows for complete healing and revascularization of the recipient site; typically three months or more. One small case series from Japan, of patients presenting with palpable indurations six months to six years post injection by “untrained individuals” reported pain, infection, breast discharge, lymphadenopathy, and cysts requiring surgical excision (16). They and others have postulated that possible large volume mass injection is a contributing factor to large areas of fat necrosis (16-18). Mammographic changes observed in breasts subjected to autologous fat transfer include various calcifications, cysts, and tissue remodeling in less than half of patients (17). It has been argued that these changes may interfere with breast cancer screening (16, 19), but it has also been argued that the incidence and significance of these findings are no worse than those following other cosmetic breast procedures (4, 20). Furthermore, it has been stated that radiographic multimodal follow-up evaluation is effective at discerning the characteristics of injected fat over time and that no increase in the increase of breast cancer has been reported to date (17, 21). SummaryBreast augmentation with autologous fat transfer is an option for some women seeking a modest increase in size and/or the correction of breast contour deformities. Partial loss of injected volume and the need for multiple treatment sessions are common. The current literature demonstrates low complication rates in experienced hands, mammographic changes distinct from cancer at a rate no higher than that of other cosmetic breast operations, and no apparent effect on the incidence of breast cancer. The use of adjuncts including external tissue expansion, adipose derived stem cells and platelet-rich plasma has not been proven superior, but has not been studied adequately. References1. Cosmetic Surgery National Data Bank. 2008 Statistics. American Society for Aesthetic Plastic Surgery 2009. P. 3.
2. Bircoll MJ. Case report: Cosmetic breast augmentation utilizing autologous fat and liposuction techniques. Plast Reconstr Surg 1987;79:267-71. 3. ASPRS Ad-Hoc Committee on New Procedures. Report on autologous fat transplantation. September 30, 1987. 4. Coleman SR, Saboeiro AP. Fat grafting to the breast revisited: Safety and efficacy. Plast Reconstr Surg 2007; 119:775-85. 5. Gutowski KA. Current applications and safety of autologous fat grafts: A report of the ASPS Fat Graft Task Force. Plast Reconstr Surg 2009;124:272-80. 6. Pelosi MA III, Pelosi MA II. Breast Augmentation. In: Rayburn WF, Laube DW, eds. Cosmetic Procedures in Gynecology. Obstetrics and Gynecology Clinics of North America 2010 December; Vol. 37, No. 4: 533-46. 7. Khoury RK, Schlenz I, Murphy BJ, Baker TJ. Nonsurgical breast enlargement using an external soft-tissue expansion system. Plast Reconstr Surg 2000;105:2500-12. 8. Khoury RK, et al. Brava and autologous fat transfer is a safe and effective breast augmentation alternative: Results of a 6-year, 81-patient, prospective multicenter study. Plast Reconstr Surg 2012;129:1173-87. 9. Mizuno H, Hyakusoku H. Fat grafting to the breast and adipose-derived stem cells: Recent scientific consensus and controversy. Aesthet Surg J 2010;30:381-9. 10. Yoshimura K, et al. Cell-assisted lipotransfer for cosmetic breast augmentation: Supportive use of adipose-derived stem/stromal cells. Aesthet Plast Surg 2008;32:48-55. 11. Fraser JK, Hedrick MH, Cohen SR. Oncologic risks of autologous fat grafting to the breast. Aesthet Surg J 2011;31:68-75. 12. http://www.selphyl.com 13. Eppley BL, Pietrzak WS, Blanton M. Platelet-rich plasma: A review of biology and applications in plastic surgery. Plast Reconstr Surg 2006;118:147e-159e. 14. Salgarello M, Visconti G, Rusciani A. Breast fat grafting with platelet-rich plasma: A comparative clinical study and current state of the art. Plast Reconstr Surg 2011;127:2176-85.) 15. Pelosi MA III, Pelosi MA II. Liposuction. In: Rayburn WF, Laube DW, eds. Cosmetic Procedures in Gynecology. Obstetrics and Gynecology Clinics of North America 2010 December; Vol. 37, No. 4: 507-19. 16. Hyakusoku H, et al. Complications after autologous fat transfer to the breast. Plast Reconstr Surg 2009;123:360-70. 17. Veber M, et al. Radiographic findings after breast augmentation by autologous fat transfer. Plast Reconstr Surg 2011;127:1289-99. 18. Delay E, Garson S, Tousson G, Sinna R. Fat injection to the breast: Technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J 2009;29:360-76. 19. Spear S, Al-Attar A. Discussion: Mammographic changes after fat transfer to the breast compared with changes after breast reduction. Plast Reconstr Surg 2012;129:1039-41. 20. Rubin JP, et al. Mammographic changes after fat transfer to the breast compared with changes after breast reduction: A blinded study. Plast Reconstr Surg 2012:1029-38. 21. Carvajal J, Patiño JH. Mammographic findings after breast augmentation with autologous fat injection. Aesthet Surg J 2008;28:153-62.
3 Comments
9/25/2020 03:31:35 am
Thank you for this informative post, and it seems quite helpful for individuals who are considering such procedures.
Reply
2/3/2023 12:39:54 am
Nice Blog! The information you have provided is incredibly detailed and insightful. We appreciate you sharing this important bit of information with us.
Reply
6/20/2023 10:17:54 pm
I read your blog post on breast augmentation with autologous fat injections with great interest. The topic of natural breast enhancement using one's own fat is indeed intriguing and has gained popularity in recent years.
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorMarco A. Pelosi, III, MD, is a cosmetic gynecologist, surgeon, lecturer & cofounder of the ISCG. You may contact him directly at [email protected] Archives
April 2020
Categories
All
|
We are ISCG |
Get to Know Us |
Stay Connected |






 RSS Feed
RSS Feed